 They were asking us that when the be a cl two when it's going to be heated in the test, we have to find out the anhydrous B A C L to the amount of the mass of that. So this is how we have been calculating the molar mass now in the next step of these questions. Okay, I can just write like this way to 46.47 grand Permal. So by rounding up those figures, We can write four 2 45 point Okay, it will be a 46945. That is grandpa mole When we are used to do the all addition and multiplication. Atomic weight of Magnesium (Mg): 24.305 (Ref: Jlab-ele012) Atomic weight of Sulfur (S): 32.066 (Ref: Jlab-ele016) Atomic weight of Oxygen (O): 15.9994 (Ref: Jlab-ele008) Step 3: Calculate the molecular weight of Magnesium sulfate by adding the total weight of all atoms. What? We're going to be closing this bracket. Step 2: Find out the atomic weights of each atom (from the periodic table). Okay, one point hydrogen is having one porn zito 0784 Brackett clothes plus oxygen is having 15.999 in this old thing out there. So we are just writing those seven outside and now to bracket. Okay, we can write like this too, is again over here. So we are going to be right over here, seven outside of the bracket. We are going to be right as now s is having a sorry, oxygen is having what? Oxygen is having 15. So we are going to be right over here, four multiplied in the back outside of the bracket. Whatever we have written just about that only we have to write it Plus this is four, right? This is four multiplied over there. 305 plus the molar mass off or we can say atomic mass off. So just simply we have to put all the value over here in this particular box brackett Mama, Llama. So we are just putting those things in the formula that is smaller Mas mole Armas of M G. So for finding out now we are writing over here the formula of moller Maas. That is MGs Oh and S these are the item and those are the atomic mass of M G. So for finding out the molar mass, we should know the atomic mass of each element which is present in the MAg nation. We have to do just a simple addition along with the multiplication. We should know about the atom and atomic mass from that. Magnesium sulfate is a magnesium salt having sulfate as the counterion. CID 5462224 (Magnesium) CID 1118 (Sulfuric Acid) Dates. Also that one mole Armas, how we are going to be finding out for that. Computed by PubChem 2.2 (PubChem release 2021.10.14) Component Compounds. So this will be about first step in the next question they were supposed to ask us, what is moral Armas Armas of this magnification self it helps to hydrate. Okay so the name itself, it's going to be re present like magnesia self it helps to hydrate. Hector is going to be used with a hydrated. So we are using prefects over here a hep to okay we are perfect, you're using prefects. So here we can say the name of this, how we can represent this as we are knowing that there are seven water molecules. So in the first step we are supposed to find out the name of this um MGs. 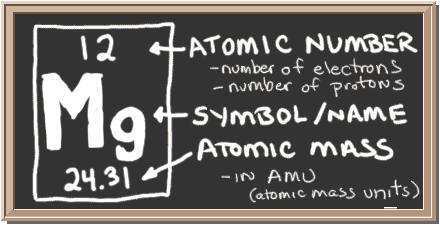
So here in the question we have been supposed to find out the things in the step vice manner. Show ALL calculations.Ī hydrate of magnesium sulfate has the molecular formula MgSO4Â♷H2O. Show ALL calculations.Ĭalculate the molar ratio of anhydrous BaCl2 and water in the hydrate, and fill in n in the following formula of the hydrate BaCl2Â Show ALL calculations.Ĭalculate the moles of BaCl2 in the sample before heating (do this by dividing the mass of anhydrous BaCl2 in the sample by the molar mass of BaCl2). Show ALL calculations.Ĭalculate the moles of water in the sample before heating (do this by dividing the mass of water in the sample by the molar mass of water). Show ALL calculations.Ĭalculate the percentage of water in the sample before heating. Mass of test tube plus solid sample after heating = 13.43 gĬalculate the mass of the hydrate before heating, the mass of water in the hydrate before heating, and the mass of anhydrous BaCl2. Mass of test tube plus solid sample before heating = 13.60 g nH2O to complete dryness in a test tube.Show ALL calculations.Ī student heated a sample of solid BaCl2Â What is the name of it?Ĭalculate the molar mass of MgSO4Â♷H2O by adding the atomic masses for all atoms present in the hydrate. And don’t forget to put the unit g/mol to your final calculated molar mass.SOLVED: A hydrate of magnesium sulfate has the molecular formula MgSO4Â♷H2O.First solve the brackets, then multiplications and at last do the final addition. Always follow the calculation order to avoid any mistakes in calculation.But all these units (i.e g/mol, grams/mole and g/mole) are the same. In some books, you may see the unit of molar mass as grams/mole or g/mole.I hope you have understood the short and simple calculation for finding the molar mass of Al2O3. Hence the Molar mass of Al2O3 is 101.961 g/mol. 
So, Molar mass of Al2O3 = Molar mass of 2 Aluminum (Al) atoms + Molar mass of 3 Oxygen (O) atoms. You can see that in Al2O3, there are 2 Aluminum atoms and 3 Oxygen atoms. Now, to calculate the molar mass of Al2O3, you just have to add the molar mass of all the individual atoms that are present in Al2O3. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
